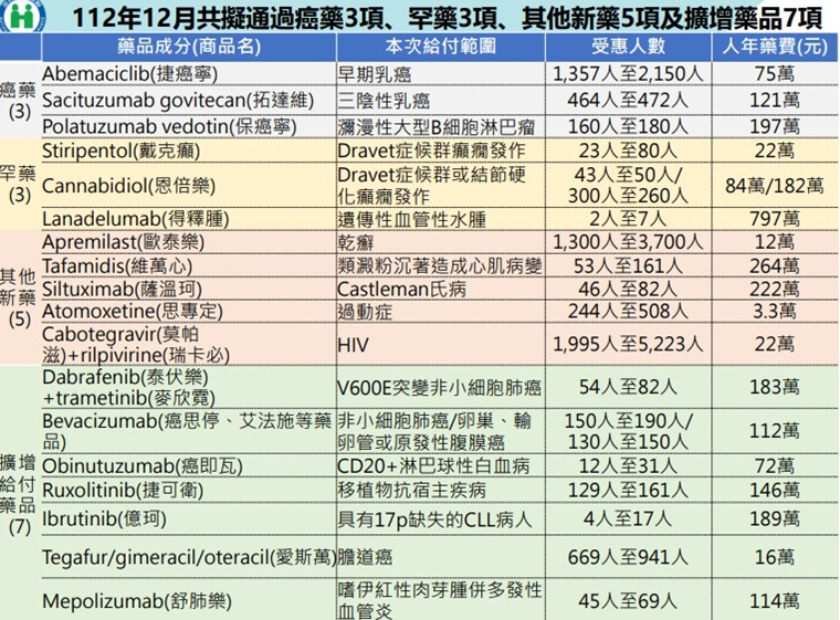
¤¤¥،°·±d«OہI¸p،u¥¥ء°·±d«OہIأؤھ«µ¹¥I¶µ¥ط¤خ¤ن¥I¼ذ·ا¦@¦Pہہq·|ؤ³،v°w¹ï¦´ءھvہّ¤خ«e½uھvہّ،A³q¹L¦h¶µ·sأؤ¯ا¤J°·«Oµ¹¥I©MآX¼Wµ¹¥I،A¥]¬Aہù¯g·sأؤھvہّ¦´ء¨إہù،B±ك´ء©خآಾ©ت¤T³±©ت¨إہùµ¥¦h؛ط¤£¦Pہù§O،F¨u¯f·sأؤ،A¦pھvہّDravet¯gش¸s،A¥H¤خ¨ن¥L·sأؤ،A¦pھvہّ°®إ~µ¥¯e¯f،A¦@p¦¬¸ü11¶µ·sأؤ¤خ7¶µأؤ«~آX¼Wµ¹¥I³W©w،Aء`¦@ر؛ھ`أؤ¶O57»ُ¤¸،A¹w¦ô¶W¹L14,554¯f¤ح¨ü´f،C
°·«O¸p¦¬¸ü¦h¶µ·sأؤ §Uہù¤ح°§C´_µo·ہI»P©µھّ¦s¬،´ء
°·«O¸pھي¥ـ،A¥»¦¸·|ؤ³¦P·N¦¬¸ü§tabemaciclib¦¨¤ہ·sہùأؤ،A¦³®ؤ°§C¦´ء
¨إہù¯f¤ح30%¥H¤W´_µo·ہI،F¦P®ة¦¬¸ü¤£¦³µLBRCA°ٍ¦]¬ًإـ¤§¤T³±©ت¨إہù±wھج§،¥i¨د¥خ¤§§tsacituzumab govitecan¦¨¤ہ¼ذ¹vأؤھ«،A¥i©µھّ¾مإé¦s¬،´ء¬ù5.4س¤ë،C
¥t¬°·سإU§َ¦hہù¯g¯f¤ح،B´î»´ہù¤ح®a®xt¾ل،A¦P·N¦¬¸üھvہّ،uؤj؛©©ت¤j«¬B²سM²O¤ع½F،v§tpolatuzumab vedotin¦¨¤ہ·sأؤ،AإµLھk¨د¥خCAR-T²سMھvہّ¤§ہù¤ح،A¦³¥t¤@سأؤ«~¥i؟ï¥خ،F¥t¦P·N§tdabrafenib¤خtrametinib¦¨¤ہأؤ«~¨ض¥خھvہّ±ك´ء«D¤p²سMھحہù،A¾مإé¦s¬،´ء¬ù18.2س¤ë،F§tobinutuzumab¦¨¤ہأؤ«~¥خ©َھvہّ²O¤ع²y©ت¥ص¦ه¯f،A¥i©µھّµL´c¤ئ¦s¬،´ء¬ù13.2س¤ë،C
¦³2¶µہùأؤ¦]¼t°س¦P·N°»ùآX¼Wµ¹¥I½d³ٍ،A¥]§tھvہّ§Z±_،B؟é§Z؛ق©خ¸،½¤ہù¤§§tbevacizumab¦¨¤ہأؤ«~¤خھvہّ¨م¦³17p¯ت¥¢ھ؛CLL¤§§tibrutinib¦¨¤ہأؤ«~،C